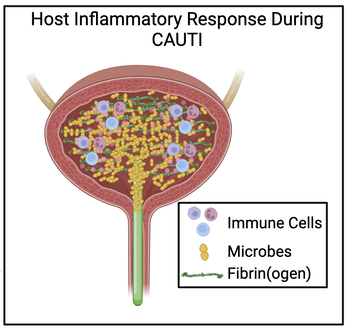
Upon urinary catheterization, tissue damage initiates the coagulation cascade to produce and release fibrinogen into the bladder where it coats the catheter providing a platform for pathogens such as E. faecalis to colonize. As part of the inflammation and infection host response, macrophages are recruited to the bladder; however, it has been found that these macrophages are not able to control infection. Since accumulated fibrinogen polymerizes to fibrin during catheterization, my project characterizes how fibrinogen and fibrin distinctly affects macrophage polarization and how its dysfunction contributes to CAUTI pathogenesis. Our project utilizes various immunological approaches, including flow cytometry, to characterize the role that fibrinogen and fibrin has on rendering macrophages and other innate immune cells ineffective at clearing CAUTI pathogens and properly orchestrating the tissue wound healing process.
Inhibition of Host-Protein Deposition on Urinary Catheters to Reduce CAUTIs
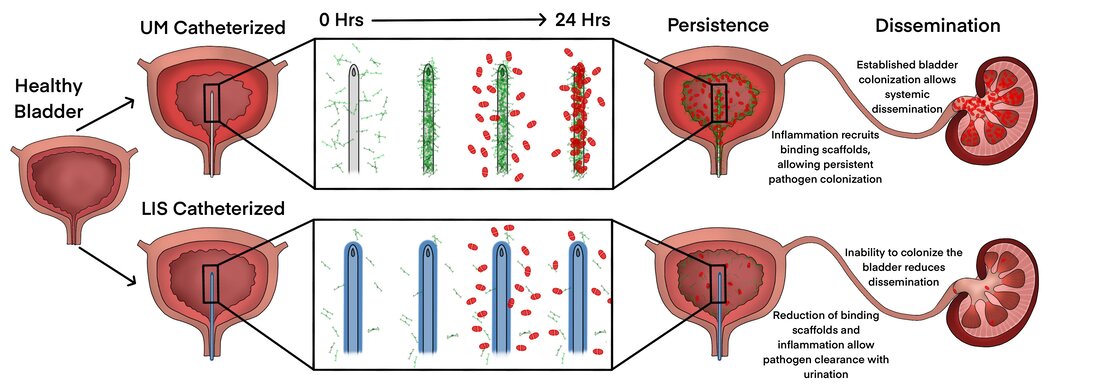
Microbial adhesion to medical devices is common for hospital acquired infections, particularly, those associated with urinary catheters. Studies show catheter use elicits bladder inflammation, releasing host serum-proteins, including fibrinogen, into the bladder, which deposit on the urinary catheter. Additionally, it’s been shown that uropathogens use fibrinogen as a scaffold to persist in the bladder and inhibition of this interaction significantly reduces infection. Thus, this project aims to target catheter protein deposition, specifically fibrinogen, to reduce colonization creating an effective antibiotic-sparring and preventative treatment against CAUTI. We have shown that deposited fibrinogen is advantageous for multiple uropathogens and in a mouse model of CAUTI a liquid-infused catheter is able to decrease protein deposition and microbial colonization of the catheter and bladder.
Candida spp are a major causative agent of CAUTI – specifically Candida albicans– that has steadily increased to become the second most common CAUTI uropathogen. Even though Candida spp are causing 17.8% of CAUTIs, there is poor understanding of the molecular details of how C. albicans attaches, grows in the bladder, forms biofilms, survives, and persists during CAUTI. As use of urinary catheters is steadily increasing and fungal infections remain difficult to treat, understanding how Candida spp establishes an infection in the catheterized patient is of utmost public health importance. This project goal is to characterize the fungal mechanisms of pathogenesis and host-fungal interaction during Candida CAUTI.
The bladder is an elastic organ of the urinary tract with innate immune defenses including an inner-most urothelial cell layer as well as resident and recruitable immune cells. These defenses are disrupted by urinary catheters which perturb bladder homeostasis by mechanically manipulating the urothelium and perpetually promoting inflammation. Aimed at healing damaged tissues, inflammation inherently recruits fibrinogen (Fg) and immune cells to promote healing. Paradoxically, Fg also endangers the catheterized bladder by loading onto catheters and creating a scaffold for bacteria to develop biofilms which promote infection; Catheter-Associated Urinary Tract Infections (CAUTIs). The microenvironment in the inflamed bladder during CAUTIs remains uninvestigated and because this inflammatory response during catheterization complements pathogenesis, I hypothesize we can mitigate CAUTIs by targeting and decreasing the inflammation. This will allow us to develop therapeutics that can be translatable to clinics and shift catheterization towards a better outcome.
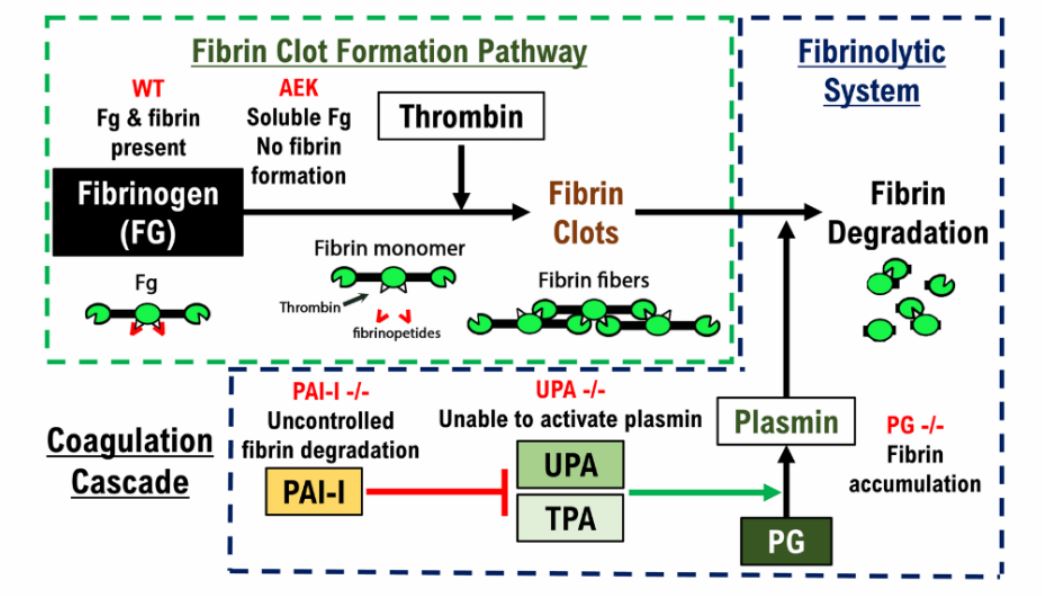
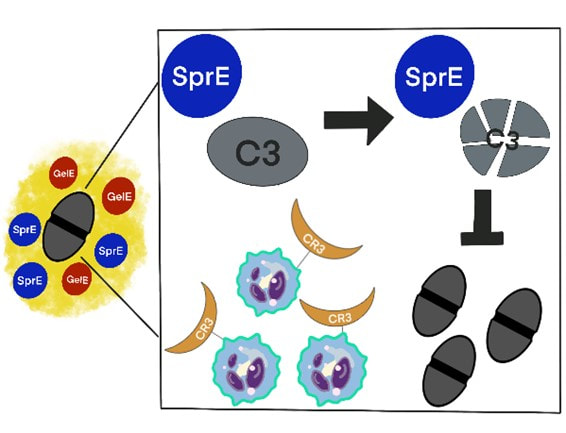
E. faecalis is one of the most prevalent causative pathogens of CAUTI representing 15% to 30% of cases. Their treatment is difficult due to intrinsic antibiotic resistances and the emergence of multidrug resistant strains. E. faecalis’s ability to form biofilms on catheters allows it to persist despite robust immune response. E. faecalis secreted proteases have also been shown to be necessary for systemic dissemination. The objective of this proposal is to understand the role of E. faecalis secreted proteases in the pathogenesis of CAUTI. Preliminary studies showed that SprE degrades host factors of the fibrinolytic system, including Pg and plasmin. While previously no significant role of SprE in immune evasion has been shown, we have found activity of SprE degrading C3 in urine conditions. The fact that E. faecalis persists in the catheterized bladder despite a robust immune response and that SprE and GelE are induced during CAUTI suggests that proteases may be important for enterococcal pathogenesis. Due to the prevalence of E. faecalis in CAUTIs, difficulty of treatment, potential for dissemination and resulting complications, it is critical to understand the role of secreted proteases in pathogenesis.
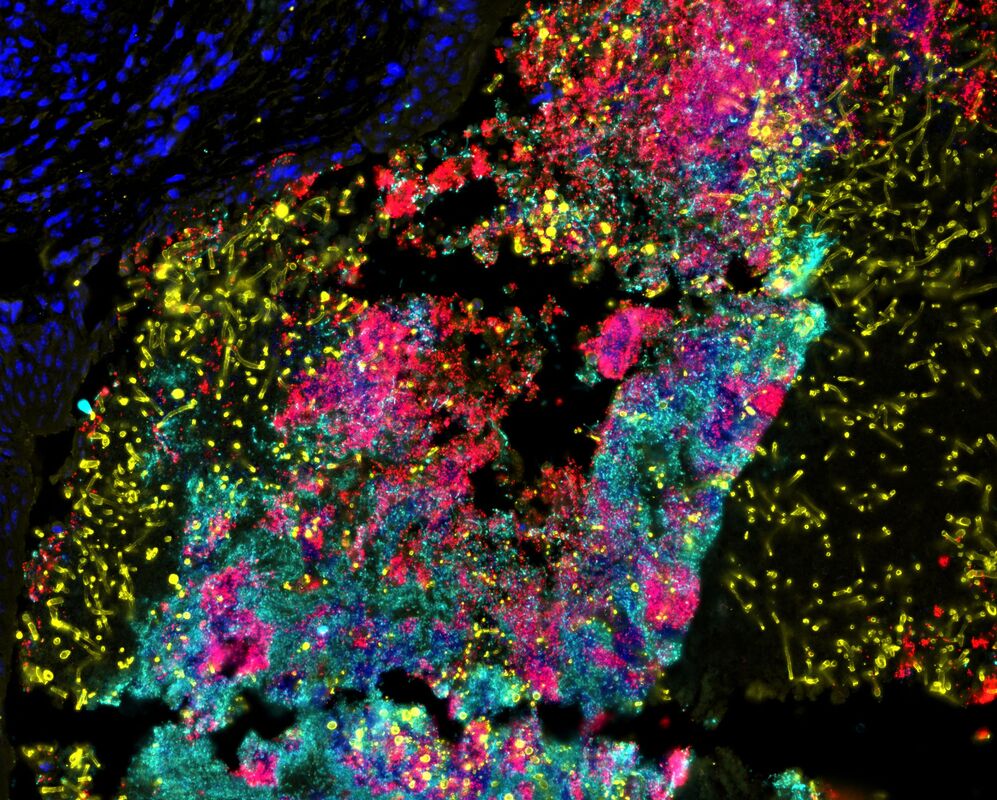
During Catheter Associated Urinary Tract Infection (CAUTI), many different pathogens colonize the bladder and the catheter including Enterococcus faecalis, Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Candida albicans. The interactions between these pathogens are not currently known. Our lab aims to understand how they interact with each other in possible co-biofilms and how fibrinogen comes into play during infections. We focus on E. faecalis as the primary agent as previous studies have suggested that E. faecalis can affect the growth of other pathogens such as E. coli and C. albicans.